About Us
AI-Powered Blood Tests for the Prevention & Early Detection of Cancers & Chronic Diseases
20/20 BioLabs (formerly 2020 GeneSystems) develops and commercializes cutting-edge, AI-powered laboratory-based blood tests for the early detection and prevention of cancers and chronic diseases. Our advanced diagnostics leverage machine learning algorithms built on extensive real-world data, offering exceptional accuracy and convenience for individuals and providers.
We pioneer affordable, accurate, actionable tests that can be conveniently accessed at home using new, upper arm collection devices that avoid painful needles.
(We call these advantages our “straight A’s)
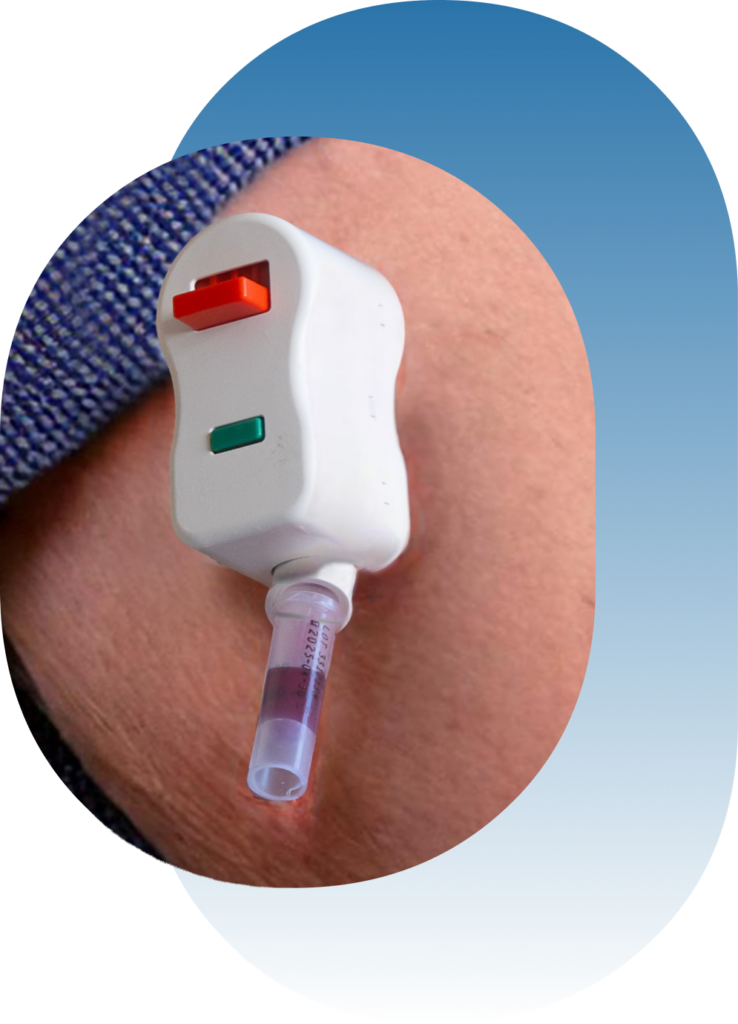
OneTest™ for Cancer
Our protein tumor marker approach is the only multi-cancer early detection (MCED) blood test that:
Detects multiple cancers including lung, pancreatic, ovarian, and liver at earlier, more treatable stages than competing, DNA based MCEDs.
Starts at under $200, versus just under $1,000 from leading competitors.
Can be collected at-home using the device painless, upper arm capillary blood collection.
Key Benefits:
AI-driven results:
Enhanced accuracy through machine learning algorithms trained on data from tens of thousands of individuals tested months or years before being diagnosed with cancer.
CAP Accredited & CLIA Licensed laboratory
ensuring the highest standards of accuracy and reliability.
Our MCED performed well in blinded studies conducted by the U.S. National Cancer Institute and our biomarker approach is supported by studies conducted by the MD Anderson Cancer Center and other leading academic medical centers.

Available now!
Introducing: OneTest for Longevity
Our newest test measures biomarkers of inflammation associated with chronic diseases including heart attacks, cancers, dementia, diabetes, and mental health. We offer specific guidance to improve diet and lifestyle to lower biomarkers and chronic inflammation to improve health aging.
Anticipated launch in Spring, supporting proactive disease prevention strategies aligned with “US Government-sanctioned initiatives”.
For big early adopter discounts:
Our Executive Team

Jonathan Cohen, President & CEO
Mr. Cohen is the founder of our company and has served as Chief Executive Officer, President, and a director since its inception. He is a co-inventor of two of our most successful products, OneTest and BioCheck, and has led their commercial launch and sales. He has spearheaded license, research, technology transfer, investment, and sales and marketing agreements with Fortune 500 companies such as Eastman Kodak, Abbott Diagnostics, Johnson & Johnson, IBM, and Ping An. Mr. Cohen has also been a leading advocate in Annapolis, MD, and on Capitol Hill on behalf of small and emerging biotechnology and diagnostics companies. Before founding our company, Mr. Cohen was patent and general counsel for Ventana Medical Systems Inc. and Oncor Inc. He has a Master of Science Degree in Biotechnology from Johns Hopkins University and a law degree from American University.

Jiming Zhou, Ph.D., Chief Operating Officer
Dr. Zhou has over 20 years of healthcare and biotech industry experience. He began his academic career as an associate professor at Sichuan University in China and conducted extensive research at the University of Iowa. He transitioned to industrial R&D in 2005, leading major pharmaceutical projects and managing multiple clinical labs. Dr. Zhou previously co-founded Firefox Pharmaceuticals and Fairfax Medical Consulting. He received his Ph.D. in Biology from Sichuan University.

Michael Leibowitz, Ph.D., Chief Scientific Officer
Dr. Lebowitz has served as Chief Scientific Officer since January 2020, previously Director of Research & Development from 2009-2012. Concurrently, he is CSO at Athanor Biosciences. He has over 30 years of research experience, including 22 years in industry and over 18 years in research management. Dr. Lebowitz has been directly involved in launching six laboratory-developed tests and establishing two CLIA-certified labs. He holds a Ph.D. from Johns Hopkins University School of Medicine, completed a fellowship in immunology there, and is an adjunct faculty member at Johns Hopkins University and the University of Maryland, Baltimore County.

Ron Baker, Chief Business Officer
Mr. Baker has extensive experience in clinical research, operations, technical sales, marketing, and business development within oncology laboratory services. Prior to joining 20/20, he held executive positions at Roche Diagnostics, Roche Clinical Labs, and SGS Life Sciences. Mr. Baker earned his BS in Biology from Loyola University.

David Lees, Director of Sales
Mr. Lees rejoined 20/20 as Director of Sales in 2023 after exceptional performance at Software AG. He previously served as Sales Manager at 20/20 and is recognized for driving significant revenue growth. Mr. Lees holds a degree in Biochemistry from Christopher Newport University.
Investors
Our mission is to revolutionize early cancer and disease detection.
Investor information can be found by clicking the button below.